The plastic term was invented by Leo Hendrik Baekeland and means molding. It means that during a temperature interval. It maintains its quality, properties of elasticity and flexibility that allow it to adapt to different shape by extrusion, injection molding, without losing their chemical, physical and mechanical properties. This process refers to the state of matter, not to the material itself.
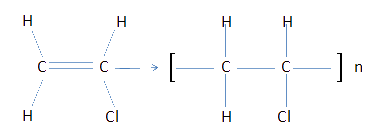
In 1930, B.F. Goodrich discovered that PVC (polyvinyl chloride) absorbs plasticizer and that when processed it results in a flexible product.
PVC is a thermoplastic addition polymer (without loss of part of the molecule) that, due to its high rigidity, makes it unwieldy, so it requires the addition of plasticizers.
It can be presented in dry white powder or paste, then called plastisol, having a temperature range between 80ºC and 140ºC to proceed with its transformation and ease of handling. Higher temperatures can start their decomposition.
PVC is degraded by the action of heat, light and electrical leakage, due to the loss of hydrochloric acid. The degraded PVC becomes rigid. Hence the convenience of using epoxidized soybean oil or other epoxidized plasticizers as secondary plasticizers.
The process from a rigid PVC to another more or less soft, is made by heating the dough and adding the appropriate plasticizers according to the application of final use.
Plasticizers should be: compatible, that is, with affinity to PVC, permanent, less volatile, low migration and low extraction and effective, what depends on the characteristics, potency and dosage.
We define plasticizer as an inert low volatility solvent of a molecular weight at least 300, which incorporated in a plastic (PVC) or synthetic rubber, increases their flexibility, elasticity and handling.
Mechanism of plasticization
The function of plasticizers is to reduce the intensity of bonds among the polymer molecules. These bonds, or attractions between molecules (Van de Waals forces), give a large rigidity to PVC.
The first plasticizer that was used was Dioctyl Phthalate (DOP) and all the studies were made from here. For this reason, was given the value one to DOP as a plasticizing power reference.
We call Substitution Factor to the amount of another plasticizer, other than the DOP, in order to plasticize equivalently, according to the following formulae:
Substitution factor (SF) = (phr plasticizer to 80/phr hardness of PDO with hardness 80).
Plasticizers can be classified according to its: effectiveness, flexibility at different temperatures, durability, continuity, composition and application. Given the number of parameters involved, we believe that can be grouped into three sections: